Description
Zobrick is a combination of Cefoperazone, a third-generation cephalosporin, and Sulbactam, a β-lactamase inhibitor. This synergistic combination is formulated in a fixed ratio to effectively treat bacterial infections, especially those caused by β-lactamase-producing resistant pathogens.
COMPOSITION
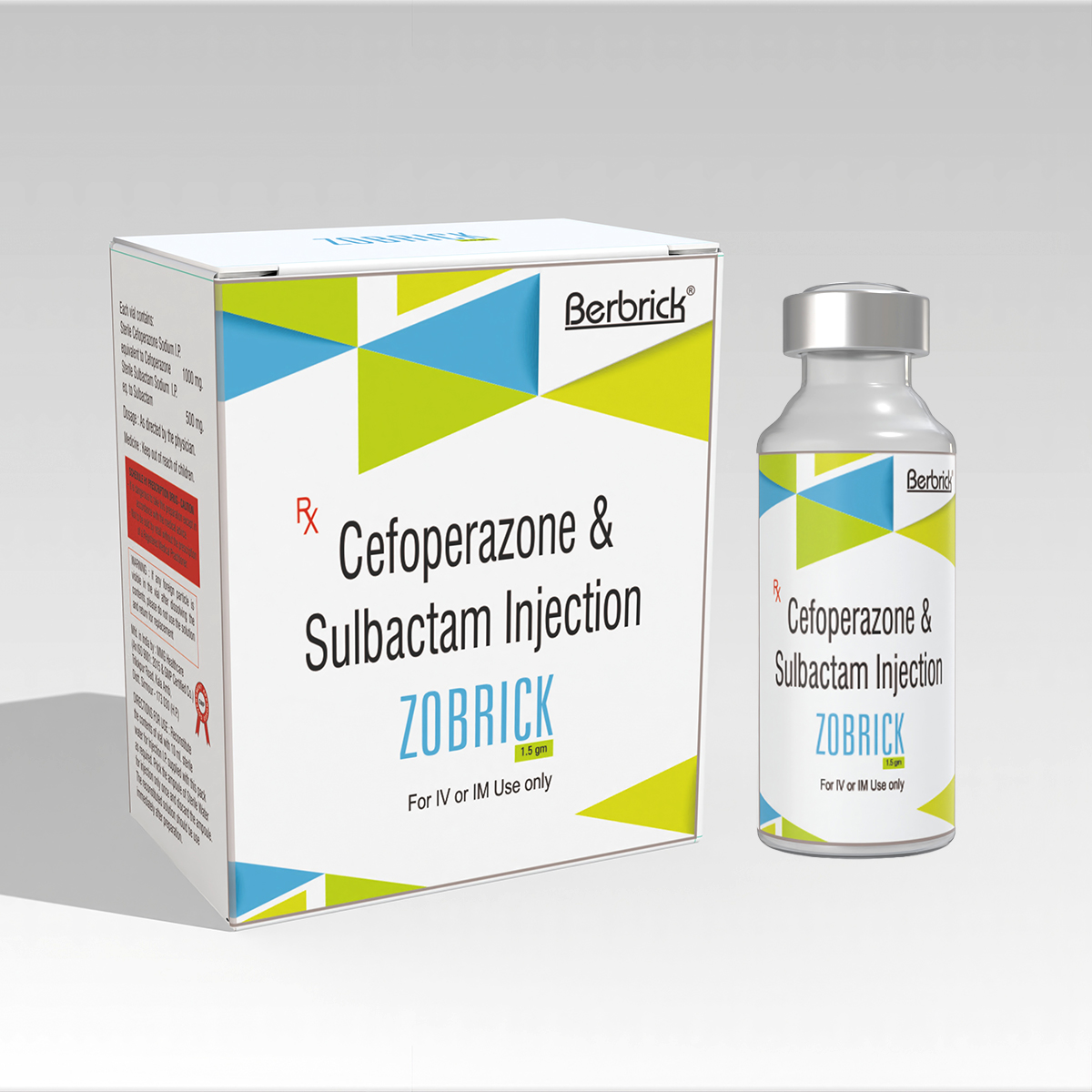
Zobrick Injection 1.5 g
Each vial contains:
- Cefoperazone Sodium equivalent to Cefoperazone IP … 1,000 mg
- Sulbactam Sodium equivalent to Sulbactam USP … 500 mg
- Sterile Water for Injection IP … 10 ml
INDICATIONS
Zobrick is indicated for the treatment of the following infections:
- Upper and lower respiratory tract infections
- Upper and lower urinary tract infections
- Intra-abdominal infections
- Septicaemia
- Meningitis
- Skin and soft tissue infections
- Bone and joint infections
- Genital tract infections
MODE OF ACTION
- Cefoperazone inhibits bacterial cell wall synthesis by blocking the formation of mucopeptides in the cell wall, causing bacterial lysis.
- Sulbactam acts as a β-lactamase inhibitor, protecting Cefoperazone from degradation and restoring its antimicrobial activity against resistant strains.
DOSAGE
Adults:
Administer in two equally divided doses per day.
- Standard dose: 3–6 g/day (Cefoperazone:Sulbactam in 2:1 ratio)
- In severe infections: Dosage may be increased up to 12 g/day (8 g Cefoperazone + 4 g Sulbactam)
- Maximum daily Sulbactam dose: 4 g
Adjust dosage in patients with renal impairment (creatinine clearance < 30 mL/min), hepatic impairment, or pediatric patients as per clinical judgement.
CLINICAL EVIDENCE
| Study | Patients | Dosage | Outcome |
| Surgical Infections (Vol. 9, No. 3, 2008) | 154 patients with intra-abdominal infections | Study Group: Cefoperazone-Sulbactam 8 g/day for 5–14 daysCompared Group: Ceftazidime 6 g/day + Amikacin 15 mg/kg/day + Metronidazole 500 mg TID | Significantly higher resolution of clinical signs & fewer adverse effects in the Zobrick group (p = 0.015) |
ADMINISTRATION
Route: Intravenous (IV) or Intramuscular (IM) use only.







Reviews
There are no reviews yet.